Cathodic Protection
- 7 October 2016
- Posted by: Stm Coatech
- Category: Educational Articles

Corrosion is an electrochemical process in which a current leaves a structure at the anode site, passes through an electrolyte, and re-enters the structure at the cathode site. For example, one small section of a pipeline may be anodic (positively charged) because it is in a soil with low resistivity compared to the rest of the line. Current would leave the pipeline at that anode site, pass through the soil, and re-enter the pipeline at a cathode (negatively charged) site. Current flows because of a potential difference between the anode and cathode. That is, the anode potential is more negative than the cathode potential, and this difference is the driving force for the corrosion current. The total system – anode, cathode, electrolyte, and metallic connection between anode and cathode is termed a corrosion cell.
Cathodic protection is a method to reduce corrosion by minimizing the difference in potential between anode and cathode. This is achieved by applying a current to the structure to be protected (such as a pipeline) from some outside source. When enough current is applied, the whole structure will be at one potential; thus, anode and cathode sites will not exist. Cathodic protection is commonly used on many types of structures, such as pipelines, underground storage tanks, locks, and ship hulls.
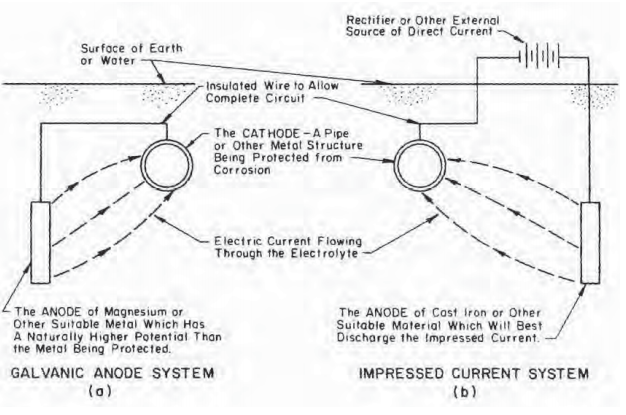
There are two main types of cathodic protection systems: galvanic and impressed current. Figure shows these two types. Note that both types have anodes (from which current flows into the electrolyte), a continuous electrolyte from the anode to the protected structure, and an external metallic connection (wire). These items are essential for all cathodic protection systems. Figure Galvanic (a) and impressed (b) current systems for cathodic protection
a.Galvanic system
A galvanic cathodic protection system makes use of the corrosive potentials for different metals. Without cathodic protection, one area of the structure exists at a more negative potential than another, and corrosion results. If, however, a much less inert object (that is, with much more negative potential, such as a magnesium anode) is placed adjacent to the structure to be protected, such as a pipeline, and a metallic connection (insulated wire) is installed between the object and the structure, the object will become the anode and the entire structure will become the cathode. That is, the new object corrodes sacrificially to protect the structure as shown in Figure (a). Thus, the galvanic cathode protection system is called a sacrificial anode cathodic protection system because the anode corrodes sacrificially to protect the structure. Galvanic anodes are usually made of either magnesium or zinc because of these metals’ higher potential compared to steel structures.
Advantages and Limitations
There are several important advantages to using galvanic anodes:
- No power source is required – Due to the fact that the protective current is generated by the electrochemical reaction between the metals, no externally supplied power is required.
- Installation and maintenance cost is reduced – Normally, galvanic anodes have the advantage of not requiring additional right-of-way purchase since the anodes are usually installed close to the protected structure. Once installed, very little maintenance is required for the life of the anode. The anode of a galvanic anode system is not subject to the same degree of electrical or mechanical malfunction as that of an impressed current system.
- Efficient and non-interfering – The relatively low, and normally well distributed, current output of the galvanic system can result in a more constant current density at the protected structure. This minimizes over protection and wasted current. The low current output reduces the possibility of interference to a minimum. The advantages mentioned enable the galvanic cathodic protection system to be utilized efficiently in a variety of applications, such as:
- For well-coated underground storage tanks and piping
- In rural areas and offshore where power is not available
- For supplemental protection, such as at pipeline crossings
- In isolated corrosive areas (hot spots)
- In highly congested, urban areas where current distribution and interference present problems
- On electrically discontinuous structures
- Well coated pipelines
However, the galvanic anode system is not without limitations. The difference in the potential of the anode and cathode that causes the protective current to flow is normally quite small. The small potential difference, or ‘driving potential’, results in very limited current outputs, especially in high soil resistivity areas. This fact severely limits the economic use of galvanic system on:
- Large structures
- Poorly-coated structures
b. Impressed current systems
Impressed current cathodic protection systems use the same elements as the galvanic protection system, only the structure is protected by applying a current to it from an anode. The anode and the structure are connected by an insulated wire, as for the galvanic system. Current flows from the anode through the electrolyte onto the structure, just as in the galvanic system. The main difference between galvanic and impressed current systems is that the galvanic system relies on the difference in potential between the anode and the structure, whereas the impressed current system uses an external power source to drive the current, as shown in Figure (b). The external power source is usually a rectifier that changes input AC power to the proper DC power level. The rectifier can be adjusted so that proper output can be maintained during the system’s life. Impressed current cathodic protection system anodes typically are high-silicone cast iron or graphite.
Advantages and Limitations
There are several important advantages to using impressed current anode systems:
- Unlimited current output capacity – The amount of current that can be designed into an impressed current system can be from a few amperes to as much as several hundred amperes.the amount of current available will be a function of the number of anodes provided, the rectifier voltage and amperage capacity and the soil resistivity in which the anodes are installed.
- Adjustable output capacity – The output of the rectifier power supply is easily adjusted to accommodate either changes in circuit resistance or current requirement. They can be provided with aotomatic control circuitry to eliminate the need for manual adjustment.
- Lower cost per ampere of cathodic protection current – galvanic anode system are significantly more expensive where amperes rather than milli-amperes of total current are required.
There are several significant disadvantages to the use of impressed current cathodic protection.
- They are more costly if only a few milliamperes of current are required to protect a small or very well coated structure. Impressed current systems typically have a base cost of several thousand dollars. If only a few galvanic anodes are required for protection of a specific structure, this will often be the more economic choice.
- Impressed current systems have a higher maintenance cost. Impressed current systems are inherently more prone to failure. Switches can be turned off and fuses can blow. Thus, the system needs to be monitored more frequently and some repair may be required once every several years.
- Impressed current systems may create stray current corrosion on other nearby structures. This is an inherent potential problem with any impressed current system. This can sometimes be minimized through the use of distributed anode designs where the anodes are placed nearby the protected structures.
REFERENCES:
1.Cathodic Protection, National Physical Laboratory, Date of access: 06 October 2016, http://www.npl.co.uk/upload/pdf/cathodic_protection_in_practise.pdf
2.Galvanic Anode Cathodic Protection System Design, Bushmann & Associates, Date of access: 06 October 2016, http://www.bushman.cc/pdf/galvanic_anode_system_design.pdf
3.Impressed Current Cathodic Protection System Design, Bushmann & Associates, Date of access: 06 October 2016, http://www.bushman.cc/pdf/impressed_current_system_design.pdf
4.Introduction to Electrical Design for Cathodic Protection Systems, Ezekiel Enterprises, Date of access: 06 October 2016, https://d6s74no67skb0.cloudfront.net/course-material/EE410-Introduction-to-Electrical-Design-for-Cathodic-Protection-Systems.pdf