Electro (Cataphoresis) Coatings
- 12 October 2018
- Posted by: Stm Coatech
- Category: Educational Articles

What is Electroplating (Cataphoresis)?
Electro-plating is a paint process that uses electricity to coat a metal part with paint. The most common metals used are steel, zinc, brass, aluminum or other conductive materials. After cleaning, the whole piece is immersed in the electrochemical cataphoresis paint tank. The dye bath contains 20% paint solids (partially remaining) and 80% deionized water.
Upon application of the electrical potential (voltage), the paint solids are transported to the surface of a molecule part at once with the electric current from the cataphoresis bath. When the outer surfaces of the part are covered, an electrical resistance is generated. As electricity always follows the least resistance, the electric current automatically flows into the areas of previously uncoated parts. This process continues until all surfaces of the part are covered with layers of the same thickness. When the entire part is coated with the correct film thickness, the flow of electricity slows down. When the electrical current is stopped, each surface in the part is coated with a solid coat of paint.
After electrostatic application of the paint solids in the cataphoresis coating, all spray and drops that will form a rough film should be removed after rinsing. The ultrafilter (UF), in which deionized water is separated from the paint solids, forms the basis of the closed-loop rinsing system. Rinse water is continuously treated and reused. Any excess non-accumulated paint can be recycled back to the paint tank.
After the spraying and drops are removed in the parts applied in the cataphoresis process, it transfers to dehydration and furnaces. The dehydration furnace is used to remove all water from the electrically applied paint film. After all the water is released, the piece enters the baking oven. The paint film in the curing oven undergoes a molecular transformation. The electrically applied paint solids are crosslinked and the acrylic modified epoxy layer is smooth and even flattened. This epoxy film is extremely dense, thin, smooth and resistant to a wide range of chemical attacks.
Cataphoresis Coating Process
Cataphoresis is a dipping coating process that is completely automatic and is based on a continuous negative electrical voltage and charging of the bath to a positive electrical voltage. It provides outstanding results and corrosion protection by drawing paint particles and obtaining a uniform paint film.
Cataphoresis coating is a cathodic process applied by dipping and gives your products very high corrosion resistance. This pioneering technology is used by car manufacturers, white goods manufacturers, producers of agricultural machinery and manufacturers of other industrial vehicles all over the world.
Cataphoresis coating systems have become universal for a modern automotive paint shop and white goods factories. Particles loaded from the dye emulsion move to the anode (AED) or cathode (CED) under electrical forces. The direct flow through the bath makes the base of the paint and pigment and resin base of the paint on the body surfaces. The coating reaches the entire recessed area. Ultrafiltration (UF) equipment is used to recover non-film-forming paints and to separate ED dye contents. After baking, the accumulated film becomes a hard, durable polymeric film.
Typical steps in electroplating for metal parts:
- 1. Cleaning (typically an alkaline cleaner, but dependent on the substrate)
2. Rinsing
3. Acid etching
4. Rinsing
5. Wetting agent dip
6. Electro coating
7. Electro coat rinsing/recovery
8. Rinsing with deionized water (DI)
9. Convection or infrared furnace curing (or both)Paint electrodes are deposited on the surface of the electrically conductive substrate to form an insulation film in the electrocoating application. As the film’s thickness increases, the conductivity of the paint surface decreases over time. At this point, the accumulated film has very little water and solvent so that it does not have a negative effect on the deposited film after rinsing with water. The coated substrate is then cooked to crosslink the polymeric film as well as to eliminate water and keep it volatile.
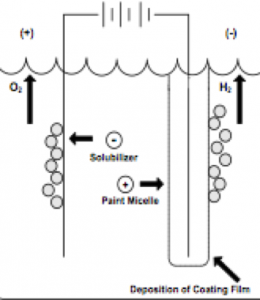
As shown in the figure below, in the cathodic electrocoating, the positively charged dye is drawn into the negatively charged cathode where the reduction occurs and causes hydrogen gas to be released. Oxidation occurs with the release of oxygen in the anode. The accumulated paint film is combined with a relatively insoluble dye film and after rinsing with one or more water, the deposited paint film enters an oven to ensure cross-linking of the cathodic paint film.
Cathodic electroplating sample
Cathode electrode deposition;
R-NH3 + + OH– -> R-NH2 + H2O
OH–ions, pH boosts and car
The electrode in the body precipitates (reducing).
Anode electrode;
H + + solvent anion -> acid
H2O is oxidized in H + form.
Many cationic epoxy electroplating resins consist of a Bisphenol A-based epoxy resin consisting of amine groups neutralized with a low molecular weight acid such as formic, acetic or lactic acid. Because it has a pH slightly below the plating bath 7, the bath components consist of stainless steel or other corrosion resistant materials to prevent rust formation.
The most common cross-linker is a blocked isocyanate, so that after the coating is cooked, the blocked isocyanate is activated and reacts with the existing hydroxyl and amine groups. Other components of a typical electroplating bath include pigment, filler pigment, water, solvent and plasticizers and low levels of modifying resins such as plasticizers, flow modifiers and catalysts.
With the film becoming more insulating as the thickness of the film approaches its maximum level, the self-coating of electro-coating paint is self-limited. Higher voltage and longer immersion times will allow higher film structures until the maximum possible film structure is reached, which is normally about 1.0 mil and 1.2 mil.
Voltage is normally 225 to 400 volts. If the voltage is too high, the coating applied to the outer surfaces will be film rupture. This is called tear stress. At an excessively high voltage, the current will pass through the film causing gas to form under the film (cathodic for anodic and hydrogen for oxygen).
Other factors affecting film structure include bath temperature and conductivity. Immersion times are normally 2-3 minutes.
- Wetting agent dip
Some electrocoating manufacturers recommend that a wetting agent be immersed in the tank just before the electro-coating tank. This is usually to prevent the bubbles from sticking to the parts as they enter the electroplating tank. Any bubble attached to the part surface prevents the accumulation of electroplating and causes a paint flaw in the finished part.
- Electro coat rinsing and recovery
An advantage of the electroplating process is the recovery of the electrocoating solid carried by the parts to the electroplating rinse tank. The electroplating rinse is continuously filtered through an ultrafiltration unit that separates the electroplating emulsion from the carrier solution and returns to the electroplating tank. Most of the electroplating is recovered in this way.
- Curing of electro coat
Since the electrocoating is essentially a liquid dye after coating a part, it will require a curing cycle depending on the type of dye chemistry used. For example, an acrylic-urethane type electrocoating will require a curing temperature of 320 F for 20 minutes (metal temperature specified for Electroclear 2800, PPG product). As with paints, the energy used to improve the electrocoating will depend on the part size and part geometry. Simple geometries can be fully improved by the use of infrared heating, because the infrared line appears and heats the metal surface without heating the entire surface. For complex geometries, a convection oven is needed to fully cure the electroplating layer in all parts of the component. Because electrocoiling chemistry may be a chemical crosslinking process, full curing requires both time and temperature to achieve optimum coating properties.
Ultrafiltration (UF) can be used to recover dye solids from deionized (DI) rinsing water, thereby reducing waste and reducing the load on waste water treatment plants. Ultrafiltration is a pressure-operated process that removes emulsifying oils, metal hydroxides, colloids, emulsions, dispersions, suspended solids and other large molecular weight substances from water and other solutions. UF membranes are characterized by molecular weight cuts.
The difficulty of many electrocoiners is to reduce DI water consumption, improve the recovery of paint solids and reduce water and wastewater treatment in electroplating applications.
Air Discharge
Electrocoat paint formulas have a slightly volatile organic compound (VOC) and hazardous air pollutant (HAP) content. Although solvent loading is greatly reduced, it will continue to be a VOC emission source as a result of the curing process. The ovens must be continuously consumed to protect the furnace atmosphere below the lower explosion limit (LEL) and to prevent smoke build-up in the factory.
The resulting exhaust contains VOCs, fumes and odor released from cured paint, which may require treatment before being released into the atmosphere. Selection of the appropriate reduction system; The safe operation of the production facility is critical for meeting the air discharge standards and for minimizing environmental odors.
In any case, the electroplating coatings have much lower VOC levels than the solvent-based spray. In addition, pollutants can be legally discharged to sewerage facilities within a few minutes, and companies are allowed to discard some VOCs into the atmosphere.
Benefits of Electro Coatings:
• Outstanding corrosion resistance
• Unchanging, double coating
• Environmentally friendly
Electroplating is environmentally friendly. Many manufacturers use electro-plating because the electroplating process produces very little HAPS (hazardous air pollutants), VOCs (volatile organic compounds) and is RoHS, OSHA and EPA compliant.
• Efficient
Almost unused paints are recycled. Most are 2 or even 2, but some are rinsing last 3, to eliminate the build-up of residual paint. The final rinses are more than filtered and any dye collected in the rinsing stages is introduced back into the application bath. Most electroplating systems are automated, the amount of paint applied to a product can be precisely controlled and different from powder or overspray paint.
• Line density
Each line is different, but with the right engineering it is possible to cover many pieces or products that can be hung simultaneously. This is based on the total frame images to be covered at once. Depending on the size of the tank, the correct number of anodes must be installed to accommodate the maximum required surface area of the tank. Unless it touches the parts, it can be filled with all tank parts. Hanging is very important for overall line efficiency.
• Compatibility
The ability to provide a very good interlayer adhesion with most topcoats allows this process to be used as a primer for almost all ferrous applications.
• Electro coating products last longer
Producers use electroplating to obtain a durable and durable finish that can withstand the chemicals used in your laundries or water and sunlight used in outdoor environments. Electrocoating is a technology used to achieve high quality, low cost finishes used worldwide. The performance advantages, along with low end-of-service costs, have made it a growing technology that will continue for a long time in the future as new uses and technologies develop.
Disadvantages:
• The construction and maintenance of an electroplating line is very expensive.
When there is suspended paint in the bathroom, it must be constantly disturbed because if the power fails, the pump will fail and the paint will be inevitable. Renovation of the paint material and the cleaning of the system may cost 10 thousand.
• Frequent discoloration is not practical.
This is not the right method to make a color specific to one or two parts. The system is installed for the continuous operation of a paint product. A few filters and settings should change, and the process of doing so is measured by days, not hours. Because there are enough materials to fill the baths, the changing material is very costly.
• UV protection.
Epoxy electro-coatings are very sensitive to sunlight. UV light breaks the coating and causes it to deteriorate. This can be overcome with a top electro-coating paint.
STM Coatech, SSPC PCI (International Coating Enspektörlüg), Turkey, Romania, Ukraine, Georgia, Russia, Azerbaijan, Turkmenistan, Kazakhstan, Iraq, Qatar, Kuwait, Oman, the Sudan and Algeria official licensors.
References:
1. Products Finishing, Electrocoating, Date of access: 24 September 2018, https://www.pfonline.com/articles/electrocoating
2. Rochester Institute of Technology, Electrophoretic Painting Process (E-Coat), Date of access: 24 September 2018, https://www.rit.edu/affiliate/nysp2i/sites/rit.edu.affiliate.nysp2i/files/pdfs/e-coating_process_final.pdf
3. Prospector, Cathodic Electrocoat: Priming the Way to Unsurpassed Product Finishing Performance, Date of access: 24 September 2018, https://knowledge.ulprospector.com/5729/pc-cathodic-electrocoating/
4. Hartford Finishing Inc., Electro Coating, Date of access: 25 September 2018, http://www.hartfordfinishing.com/e-coating/
5. Precious Electrochem, Electrophoretic, Date of access: 25 September 2018, http://www.preciouselectrochem.com/Data/TDSELECTROPHORETIC/TDSELECTROPHORETIC.pdf